 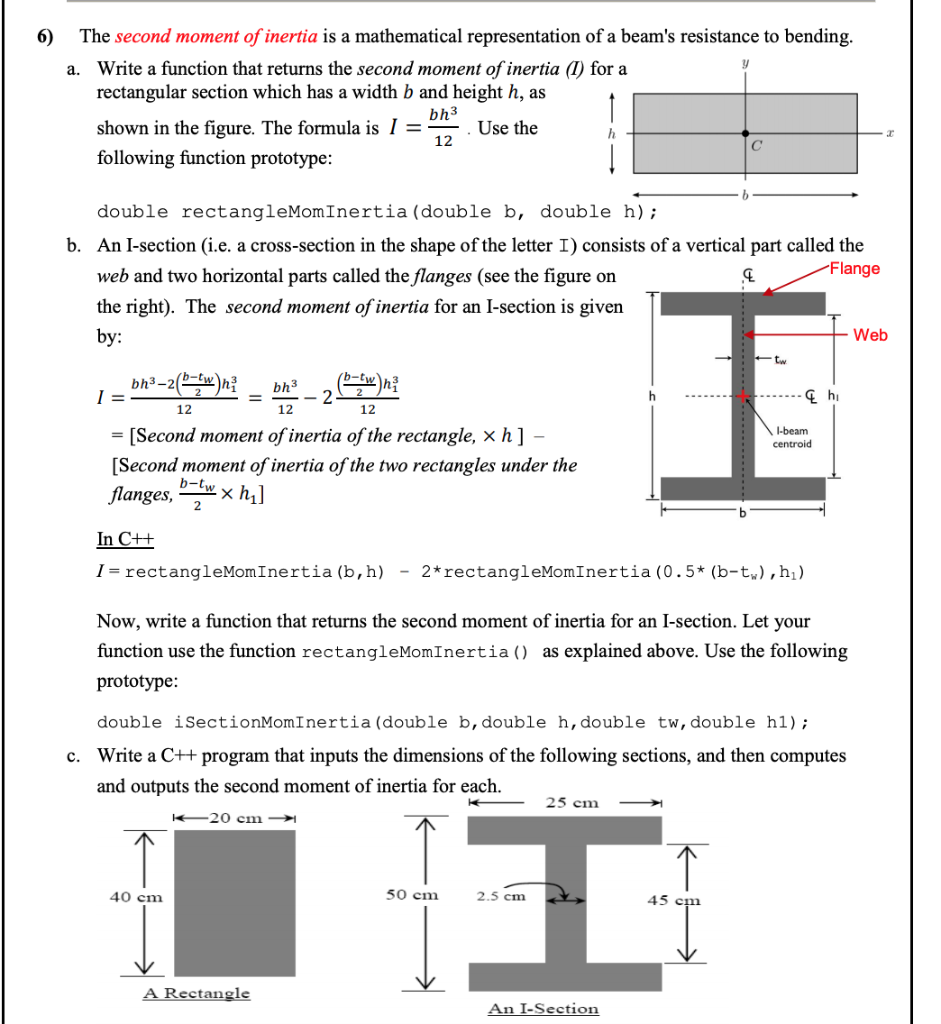 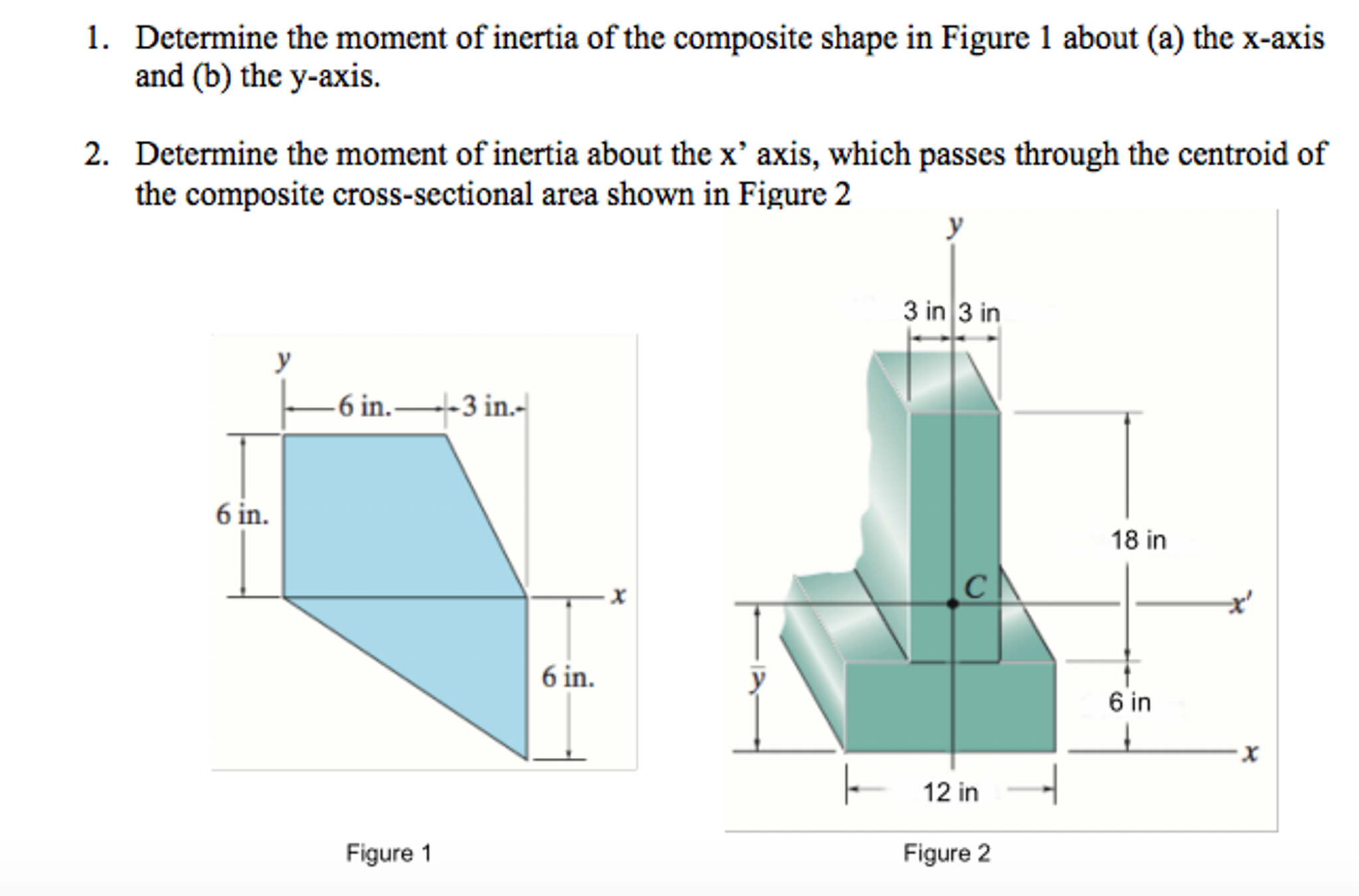
The distance of each piece of mass dm from the axis is given by the variable x, as shown in the figure. We can therefore write dm = \(\lambda\)(dx), giving us an integration variable that we know how to deal with. Note that a piece of the rod dl lies completely along the x-axis and has a length dx in fact, dl = dx in this situation. A classic among molecular spectra, the infrared absorption spectrum of HCl can be analyzed to gain information about both rotation and vibration of the molecule. This symmetric matrix is diagonalized, yielding the principal moments (the eigenvalues ) and a matrix ( ), which is made up of the normalized eigenvectors of. Mass of Cl atom 35.55×10 27kgmass of H atom 1.66×10 27kgTotal mass of the molecule 37.21×10 27 kgDistance between the two atoms 10 10 mDistance from the axis of rotation, d 210 10d0.5×10 10 mMoment of inertia, Imr 2I37.21×10 27×(0.5×10 10) 2kg.m 29.3025×10 47 kg.m 2. We chose to orient the rod along the x-axis for convenience-this is where that choice becomes very helpful. Next we have to calculate the moments of inertia (the diagonal elements) and the products of inertia (off diagonal elements) of the moment of inertia tensor ( ). If we take the differential of each side of this equation, we find 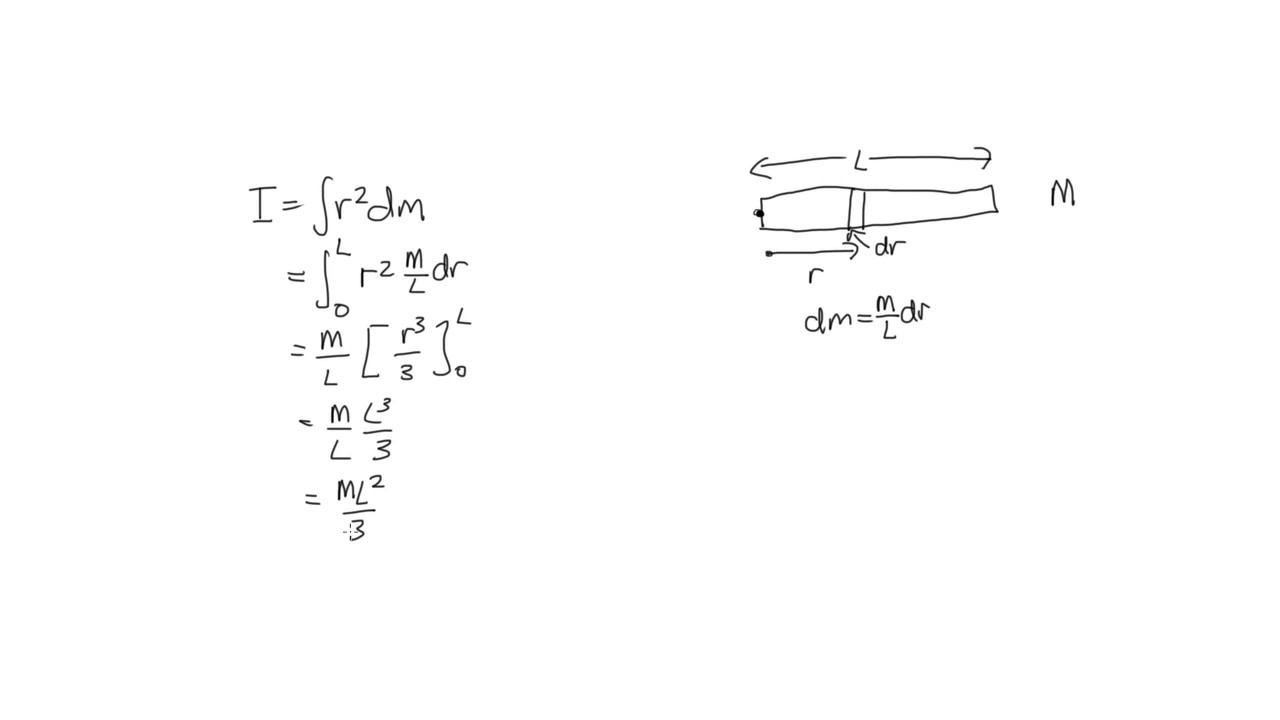 
From this result, we can conclude that it is twice as hard to rotate the barbell about the end than about its center. Of the molecules listed, HCl, ClO and CN all have permanent electric dipole moments. This movement can be completely described by three orthogonal vectors and thus contains 3 degrees of freedom.\ or\ m = \lambda l \ldotp\] In the case with the axis at the end of the barbellpassing through one of the massesthe moment of inertia is I2 m(0)2 + m(2R)2 4mR2. 10.3 Calculating moments of inertia (on p. Calculate moment of inertia of this molecule, and then use it to calculate the energies of the AEo-1 and AE2 transitions for both the 2D and 3D rotor in J. These entail the movement of the entire molecule’s center of mass. (c) (2 points) The bond distance for HCl is 1.29 A. Translational: These are the simplest of the degrees of freedom.These degrees of freedom can be broken down into 3 categories 3. This distribution makes it so that the absorption peaks that correspond to the transition from the energy state with the largest population based on the Boltzmann equation will have the largest absorption peak, with the peaks on either side steadily decreasing.Ī molecule can have three types of degrees of freedom and a total of 3N degrees of freedom, where N equals the number of atoms in the molecule. Use the proportionality constant n (2.831021) T(3/2). Calculate the moment of inertia for HCl molecule from the given value of rotational constant, B 10.40 cm. The difference in the energies of the peaks can be found by taking the difference of the frequencies of the emitted photons as delta f6106 Hz. This distribution of energy states is the main contributing factor for the observed absorption intensity distributions seen in the microwave spectrum. Use the calculated Req to solve for the moment of inertia of HCl. n 0 is the total number of molecules in the sample. Calculated geometry Rotational constant Moments of inertia Products of moments of inertia BSE Bond lengths Show me a calculated geometry.n(J) is the number of molecules in the J rotational level, and.Where E rot (J) is the molar energy of the J rotational energy state of the molecule, 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
